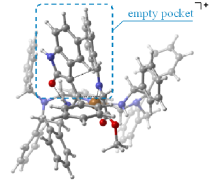
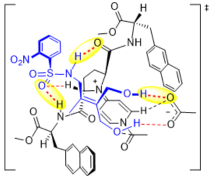
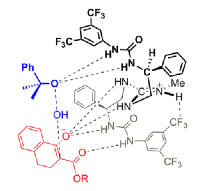
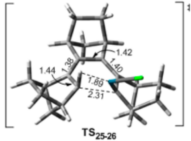
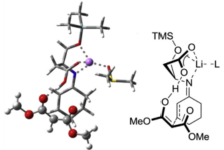
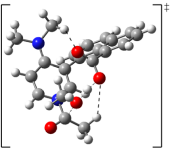
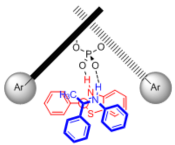
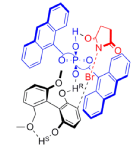
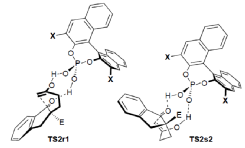
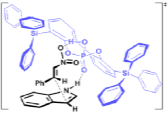
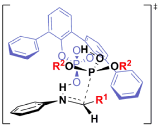
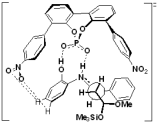
"Chiral Zn(II)-Bisamidine Complex as a Lewis-Bronsted Combined Acid Catalyst:
Application to Asymmetric Mukaiyama Aldol Reactions of a-Ketoesters"
Gotoh, R.; Yamanaka, M.
Molecules 2012, 17, 9010-9022.